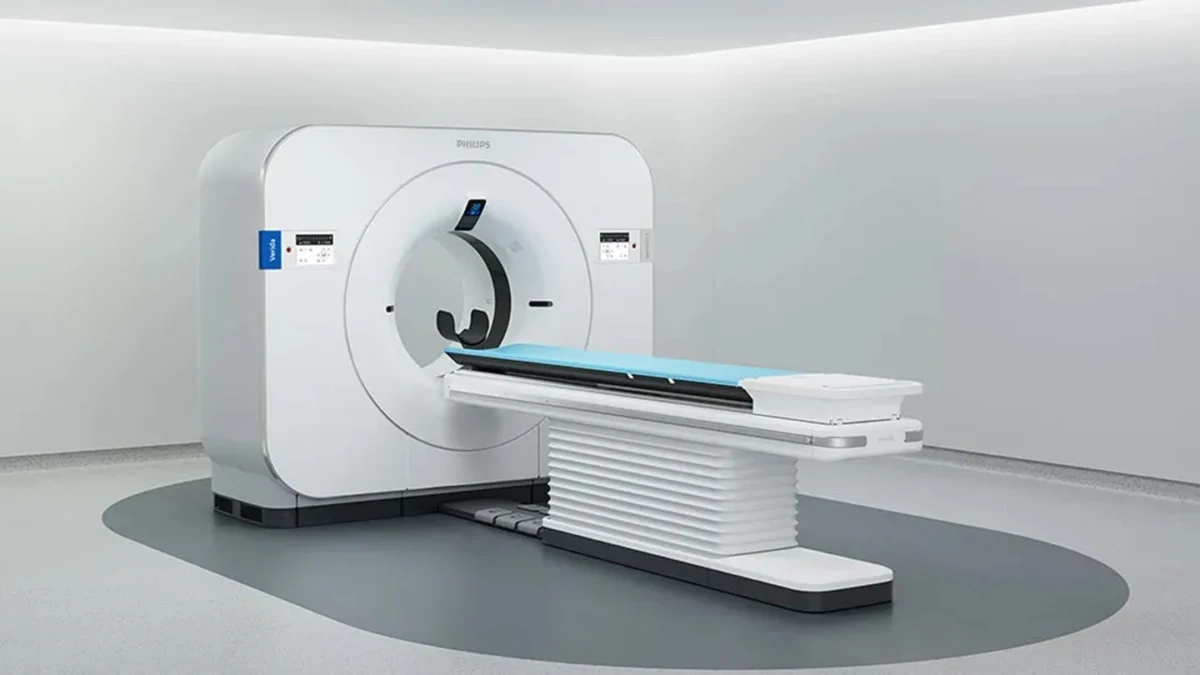
The healthcare technology landscape has reached a significant milestone with the US Food and Drug Administration (FDA) granting 510(k) clearance to Philips for its Verida system, a next-generation, AI-powered detector-based spectral computed tomography (CT) platform. This regulatory milestone signals a pivotal shift in how diagnostic imaging is performed, moving away from traditional single-energy scans toward a future where spectral data is captured intrinsically in every procedure. By integrating advanced artificial intelligence with high-performance hardware, Verida aims to solve some of the most persistent challenges in radiology, including diagnostic uncertainty, patient throughput bottlenecks, and the complexity of multi-energy imaging protocols.
The Technological Core: AI and Dual-Layer Detection
At the heart of the Verida system lies the third-generation Nano-panel Precise dual-layer detector. Unlike conventional CT detectors that capture a single energy spectrum, this dual-layer technology allows for the simultaneous acquisition of high-energy and low-energy X-ray data. This "always-on" spectral capability is a hallmark of Philips’ approach to CT, ensuring that spectral information is available for every patient without requiring the radiologist or technician to pre-select a specific mode.
To manage the massive influx of data generated by these dual-layer detectors, Philips has integrated the Spectral Precise Image technology. This suite is powered by an AI-based deep learning reconstruction engine designed specifically to address the inherent trade-off between image noise and radiation dose. In traditional imaging, reducing noise often requires increasing the radiation dose to the patient. Verida’s AI engine, however, utilizes neural networks trained on vast datasets of high-quality images to intelligently filter out noise while preserving fine structural details. This allows clinicians to customize de-noising levels based on specific clinical needs or personal preferences, ensuring that image quality remains consistent even in low-dose scenarios.
Redefining Clinical Throughput and Workflow
One of the most significant barriers to the widespread adoption of spectral CT has historically been the impact on clinical workflow. In many older systems, spectral imaging required separate scans, specialized protocols, or lengthy post-processing times, which discouraged its use in high-volume settings like emergency departments. Verida eliminates these hurdles by making spectral imaging an uninterrupted part of the standard scanning process.
The system’s performance metrics are tailored for the demands of modern, high-capacity medical facilities. Verida is capable of reconstructing 145 images per second, a speed that allows entire examinations to be processed and available for review in under 30 seconds. This rapid processing is supported by an updated computing infrastructure designed to handle the heavy computational load of AI reconstruction without lagging.
For healthcare providers, this efficiency translates into tangible productivity gains. The system is engineered to support up to 270 exams daily, making it one of the most productive imaging assets in a hospital’s portfolio. By capturing both conventional and spectral results in a single scan, the system reduces the need for "repeat and retrieve" scenarios—where a patient might otherwise have to be brought back for a second scan because the initial conventional scan was inconclusive.
Clinical Versatility and Diagnostic Precision
The clinical utility of the Verida system spans a wide array of medical disciplines, from routine screenings to complex interventional procedures. It is indicated for use in head, whole-body, cardiac, and vascular applications across all patient demographics, including pediatric and geriatric populations.
In oncology, the ability to perform material differentiation—such as distinguishing between iodine and calcium—is invaluable. Spectral CT allows clinicians to generate virtual monoenergetic images, which can enhance the visibility of hypervascular tumors or reduce the artifacts caused by metal implants. This leads to more accurate staging and treatment monitoring.
In cardiology and vascular imaging, Verida’s high-speed reconstruction and spectral data help in characterizing plaques and assessing blood flow with greater clarity. Furthermore, the system’s suitability for low-dose CT lung cancer screening and radiation therapy planning positions it as a versatile tool for early detection and precision treatment. By providing "first-time-right" imaging, Verida assists clinicians in making faster, more informed decisions, which is critical in acute care settings where every second counts.
Contextual Background: Philips’ Strategic AI Evolution
The clearance of the Verida system is not an isolated event but rather the latest step in Philips’ broader strategy to integrate artificial intelligence across its entire diagnostic and interventional portfolio. This move follows the recent FDA clearance of DeviceGuide, an AI-based software designed to assist physicians during complex mitral valve repair procedures.
Philips has been a pioneer in spectral CT technology for over a decade, starting with the launch of the IQon Spectral CT. The Verida system represents the maturation of this technology, moving it from a specialized niche tool into a mainstream workhorse. The industry trend is increasingly leaning toward "intelligent" hardware—devices that do not just capture data but also interpret and optimize it in real-time to reduce the burden on human operators.
Dan Xu, the CT business leader at Philips, emphasized this evolutionary leap during the announcement. According to Xu, the FDA clearance allows Philips to bring the next generation of spectral CT to a broader market. He noted that by combining "always-on" spectral imaging with AI-powered reconstruction, the system enables clinicians to see more clearly and act more decisively, ultimately expanding the role of CT across various clinical pathways.
Market Analysis and Industry Implications
The introduction of Verida comes at a time when the global medical imaging market is facing a dual crisis: an aging population requiring more diagnostic services and a significant shortage of qualified radiologists and technicians. Burnout in the radiology department is often linked to the high volume of scans and the complexity of modern imaging software.
Verida addresses these systemic issues by automating the most labor-intensive parts of the imaging chain. The AI-driven reconstruction reduces the time spent on manual adjustments, while the "always-on" spectral feature ensures that the necessary data is always available, preventing the administrative and clinical headaches of rescheduling patients for specialized scans.
From a competitive standpoint, Philips is positioning itself against other imaging giants such as Siemens Healthineers, GE HealthCare, and Canon Medical Systems. While competitors have their own versions of dual-energy or photon-counting CT, Philips’ focus on a detector-based spectral approach—where the separation of energies happens at the detector level rather than the source—offers specific advantages in terms of temporal alignment and cross-departmental consistency.
Financial and Operational Impact for Healthcare Systems
For hospital administrators, the investment in a system like Verida is justified by the potential for improved operational efficiency and reduced total cost of ownership. While the initial capital expenditure for AI-integrated spectral CT is higher than that of conventional CT, the long-term savings are found in several areas:
- Reduced Contrast Media: Spectral imaging can often achieve high-quality results with lower volumes of contrast agents, which is safer for patients with renal issues and reduces pharmaceutical costs.
- Decreased Follow-up Imaging: By providing definitive answers during the first scan, hospitals can reduce the number of expensive follow-up MRIs or PET scans.
- Increased Patient Throughput: The ability to handle 270 exams a day allows facilities to clear backlogs and increase revenue through higher procedure volumes.
- Enhanced Staff Retention: Simplified workflows and faster processing times can help mitigate the stresses that lead to staff turnover in radiology departments.
Chronology of Recent Milestones
To understand the trajectory of Philips’ imaging division, a look at the recent timeline of regulatory and product milestones is essential:
- Mid-2023: Philips intensifies its focus on AI-driven informatics, launching several cloud-based tools to streamline radiology data management.
- Late 2023: The company unveils the latest iterations of its dual-layer detector technology at major international radiology conferences, signaling the upcoming Verida platform.
- March 2024: Philips receives FDA clearance for DeviceGuide, proving the efficacy of its AI algorithms in live interventional cardiology settings.
- April 2024: The FDA grants 510(k) clearance for the Verida system, officially allowing its commercial distribution and clinical use in the United States.
Future Outlook: The Path to Predictive Diagnostics
The FDA clearance of Verida is likely a precursor to further innovations in "predictive" imaging. As AI algorithms continue to evolve, future updates to the Verida platform could potentially include automated lesion detection, automated measurement of organ volumes, and even the ability to predict patient outcomes based on spectral tissue signatures.
The move toward detector-based spectral CT also sets the stage for the eventual transition to photon-counting CT, which is widely considered the next "holy grail" of medical imaging. By establishing a robust AI and spectral infrastructure now with Verida, Philips is ensuring that healthcare providers are equipped with a future-proof platform that can adapt to the rapidly changing technological landscape.
In conclusion, the Verida system represents more than just a new piece of medical hardware; it is a comprehensive solution to the modern challenges of diagnostic medicine. By marrying the physical precision of dual-layer detectors with the digital intelligence of deep learning, Philips has created a tool that promises to enhance diagnostic confidence, streamline hospital operations, and ultimately improve the quality of care for patients across a multitude of clinical disciplines. As the system begins its rollout in the US market, the medical community will be watching closely to see how these theoretical efficiencies translate into real-world clinical success.