The human papillomavirus (HPV) vaccine represents a historic milestone in preventative medicine as the first and only immunization specifically designed to protect against several types of cancer. Since its introduction to the United States market in 2006, the vaccine has transitioned from a breakthrough medical innovation to a cornerstone of adolescent public health. By targeting the high-risk strains of HPV responsible for the vast majority of cervical, oropharyngeal, and anal cancers, the vaccine offers a pathway toward eliminating diseases that have claimed the lives of thousands of men and women annually. However, despite its proven efficacy and safety profile, the trajectory of HPV vaccination in the U.S. is currently defined by a complex intersection of clinical success, persistent demographic disparities, and significant recent shifts in federal health policy.
The Clinical Burden of HPV and Associated Cancers
Human papillomavirus is the most prevalent sexually transmitted infection (STI) in the United States. Epidemiological data indicates that approximately 42.5 million Americans are currently infected, with at least 13 million new infections occurring each year. While the majority of the more than 200 known HPV strains are cleared by the immune system without clinical intervention, persistent infection with high-risk oncogenic strains can lead to cellular mutations and malignancy.
The burden of HPV-related disease has shifted significantly over the last two decades. Between 2018 and 2022, more than 49,000 individuals in the U.S. developed an HPV-related cancer, a substantial increase from the 30,000 cases recorded in 1999. This rise is largely driven by a surge in oropharyngeal and anal cancers, even as cervical cancer rates have seen modest declines due to improved screening and early vaccination efforts.

Cervical Cancer and Racial Disparities
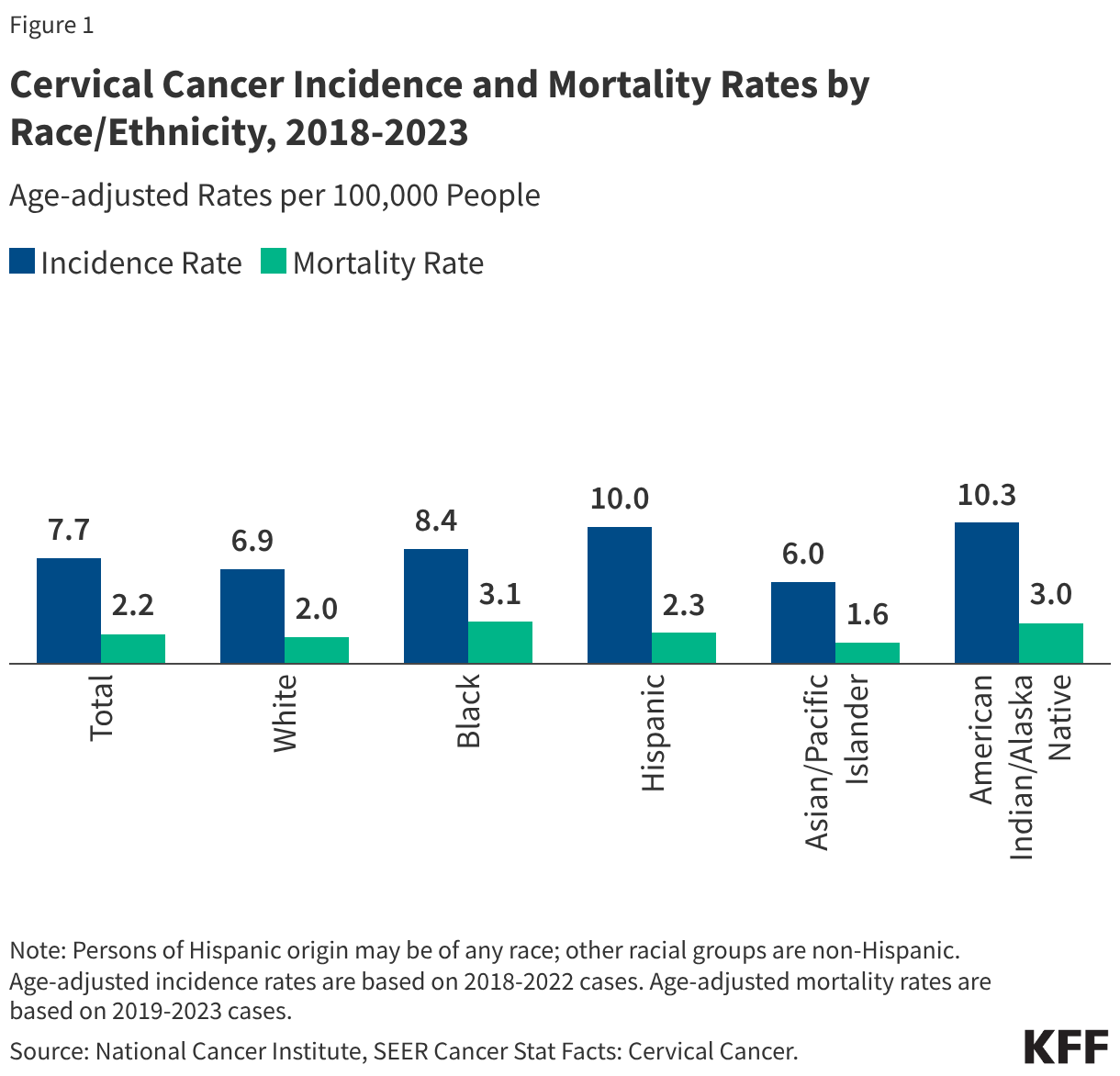
Cervical cancer remains the most prominent disease associated with HPV, with over 90% of cases linked to the virus. Strains 16 and 18 alone account for approximately 66% of cervical cancer cases globally. In 2025, it is estimated that 13,360 new cases were diagnosed in the U.S., resulting in roughly 4,320 deaths.
The impact of cervical cancer is not felt equally across all populations. Despite the widespread availability of Pap tests and HPV DNA testing, significant racial and ethnic disparities persist in mortality rates. Black women, for instance, experience the highest mortality rates from cervical cancer in the U.S., despite having the third-highest incidence rate. Hispanic women face the second-highest incidence rate but have mortality rates closer to the national average. Research suggests these outcomes are not merely biological but are rooted in systemic factors, including lower rates of follow-up treatment after abnormal screenings, diagnosis at later stages of disease progression, and historical mistrust or negative experiences within the medical system.
The Rise of Oropharyngeal and Anal Cancers
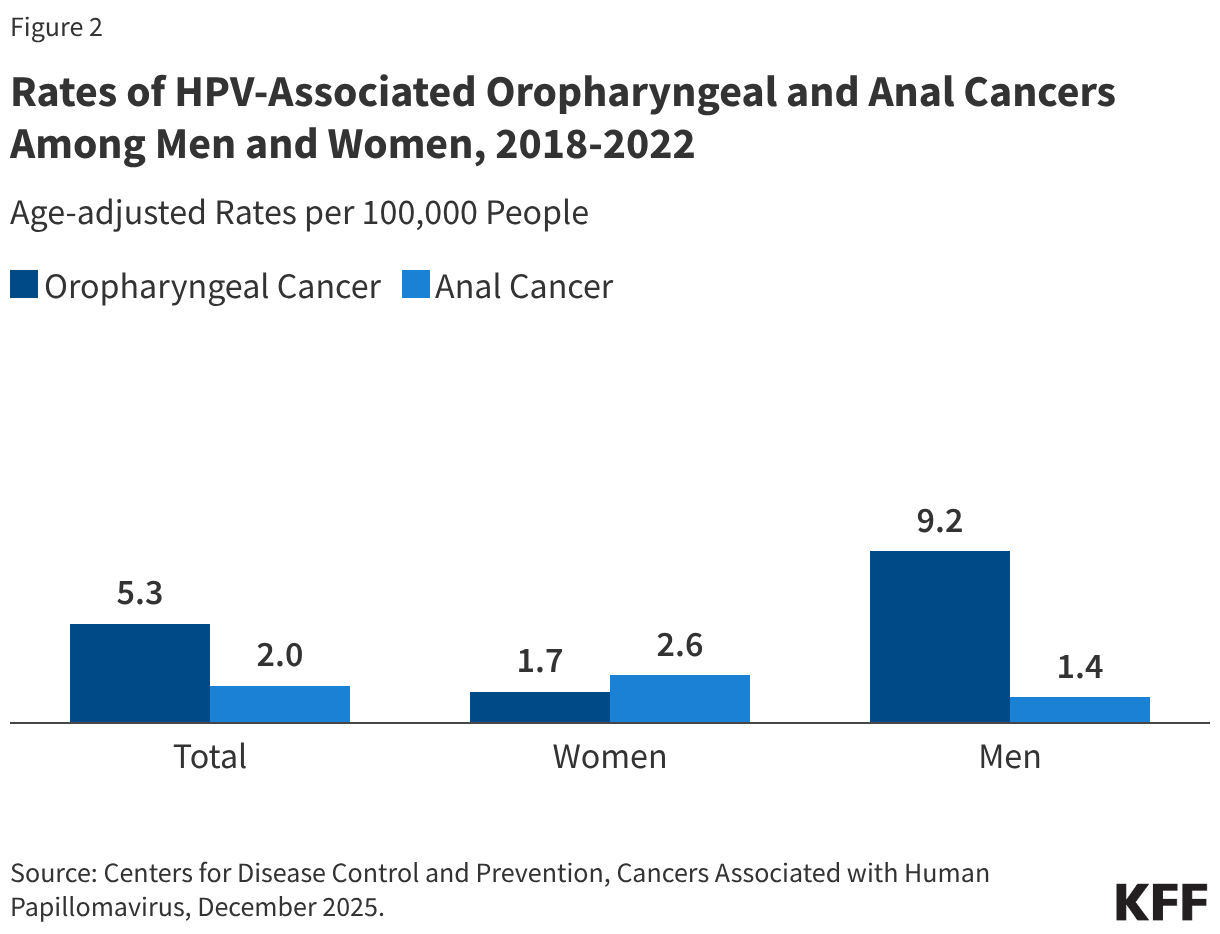
While cervical cancer is often the focus of HPV prevention, oropharyngeal (throat) cancer has emerged as the most common HPV-associated cancer among men. Approximately 22,585 cases occur annually in the U.S., with 70% attributed to HPV infection. Men are significantly more likely than women to develop these cancers, particularly those who use tobacco and alcohol heavily.
Anal cancer rates have also increased over the past 15 years. HPV is responsible for 91% of the estimated 7,600 annual cases in the U.S. While incidence is higher among women in the general population, men who have sex with men (MSM) and individuals with suppressed immune systems are at a disproportionately high risk. The expanding scope of the HPV vaccine to cover these conditions underscores its importance for individuals of all genders.
A Chronology of Vaccine Evolution and Policy Shifts
The history of the HPV vaccine in the U.S. is a timeline of expanding coverage and refined administration. The first-generation Gardasil vaccine, manufactured by Merck, was approved by the FDA in 2006 to protect against four strains (6, 11, 16, and 18).
2011–2014: Expanding the Scope
In 2011, recommendations were broadened to include boys and young men, moving away from the initial "girls-only" focus. By December 2014, the FDA approved Gardasil 9, which covers nine strains of the virus. This updated formulation protects against the strains responsible for the majority of cervical, anal, and throat cancers, as well as genital warts.
2016–2020: Refinement and Approval Expansion
By 2016, Gardasil 9 became the only HPV vaccine available in the U.S. During this period, the recommended dosage for younger adolescents was reduced from three shots to two, simplifying the schedule and improving completion rates. In 2020, the FDA further expanded the vaccine’s indicated use to include the prevention of oropharyngeal and other head and neck cancers, reflecting the changing epidemiology of HPV-related diseases.
2024–2025: Regulatory Upheaval
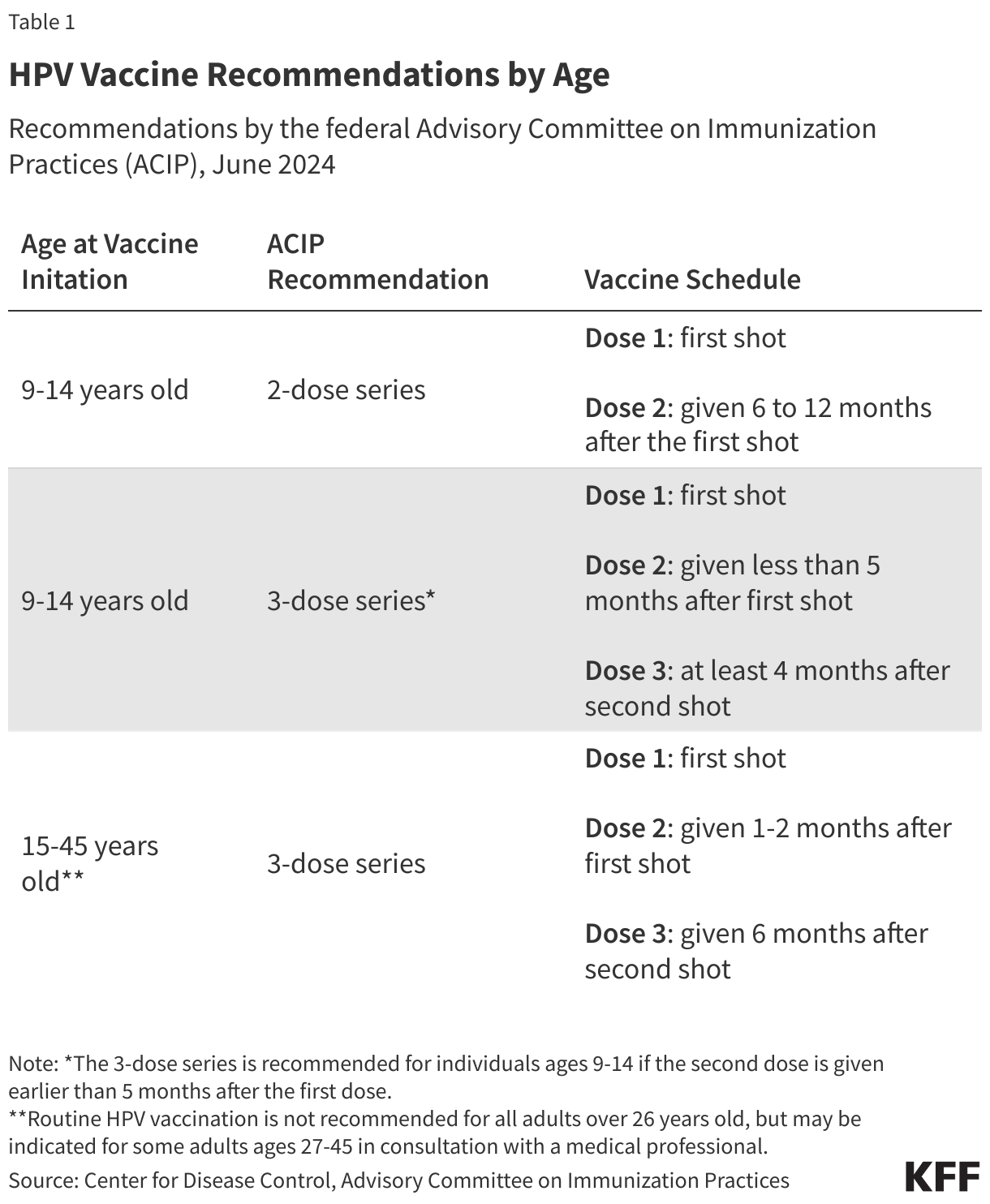
The most dramatic shifts in HPV vaccine policy occurred following the transition between the Biden and second Trump administrations. In June 2024, the federal Advisory Committee on Immunization Practices (ACIP), under the Biden administration, reaffirmed a two-dose recommendation for most adolescents, emphasizing the importance of vaccination before the onset of sexual activity.
However, in June 2025, following the inauguration of the second Trump administration, Secretary of Health and Human Services (HHS) Robert F. Kennedy Jr. dismissed the entire membership of the ACIP. The committee was reconstituted with new advisers, many of whom had publicly expressed skepticism regarding traditional vaccine schedules. In December 2025, this new committee moved to change the adolescent recommendation from two doses to a single dose. This move was immediately met with legal challenges from the American Academy of Pediatrics and other public health organizations. A federal court subsequently blocked these changes, effectively reinstating the 2024 recommendations for the time being.
Supporting Data on Vaccine Efficacy and Uptake
Global and domestic data continue to support the high efficacy of the HPV vaccine. In Scotland, where a robust bivalent vaccine program began in 2008, researchers have documented zero cases of cervical cancer among women who were fully vaccinated at ages 12 or 13. Similarly, data presented by the American Society of Clinical Oncology (ASCO) indicates that the vaccine has reduced the risk of all HPV-associated cancers in men by 50%.
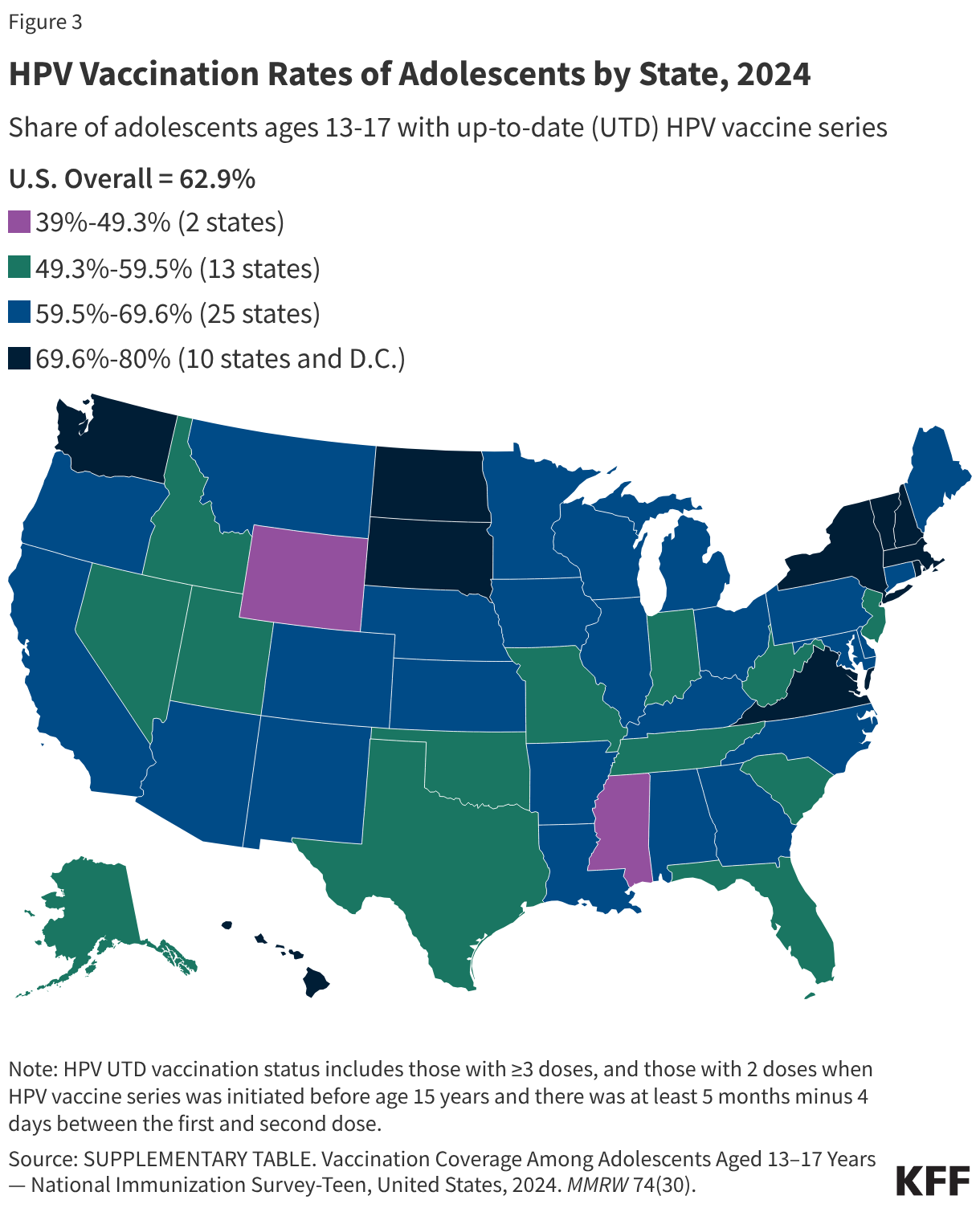
Current Vaccination Rates in the U.S.
As of 2024, approximately 60% of U.S. adolescents aged 13–17 were considered "up-to-date" (UTD) with their HPV vaccinations. While this represents progress from the vaccine’s introduction, rates have plateaued in recent years.
- Demographic Trends: Asian and Black adolescents, as well as those covered by Medicaid, are more likely to be UTD compared to White, privately insured, or uninsured adolescents.
- Gender Gap: Vaccination rates among boys (61%) remain slightly lower than among girls (64%), though the gap has narrowed since 2016.
- Geographic Variation: Success varies wildly by state, with Massachusetts reaching an 80% UTD rate, while Mississippi remains at a low of 39%.
The Rural-Urban Divide and Vaccine Hesitancy
A persistent challenge in HPV vaccine uptake is the disparity between urban and rural areas. Adolescents in rural communities continue to have lower initiation and completion rates than their urban counterparts. This is often attributed to limited access to healthcare providers and higher levels of vaccine hesitancy.
According to the CDC, the top reasons cited by parents for not vaccinating their children include safety concerns and the perception that the vaccine is unnecessary. The COVID-19 pandemic exacerbated these issues, leading to a general increase in vaccine mistrust. Public health experts express concern that the recent political volatility surrounding the ACIP and conflicting federal recommendations may further confuse parents and clinicians, potentially leading to a further decline in vaccination rates.
Vaccine Financing and Access Frameworks
To ensure broad access, the U.S. utilizes a multi-tiered financing system that effectively eliminates out-of-pocket costs for most families. The Affordable Care Act (ACA) mandates that private insurance plans cover ACIP-recommended vaccines without consumer cost-sharing.
Public Programs
Several federal and state programs provide a safety net for those without private insurance:
- Vaccines for Children (VFC): This CDC-managed program provides free vaccines to children who are Medicaid-eligible, uninsured, or underinsured.
- Medicaid and CHIP: Medicaid covers recommended vaccines for all enrollees under 21 through the EPSDT program, and recent mandates ensure coverage for adults 21 and older without cost-sharing.
- Section 317 Grants: These funds help states provide vaccines to uninsured adults who do not qualify for other programs.
- Manufacturer Assistance: Merck operates a Patient Assistance Program for Gardasil 9, providing the vaccine at no cost to low-income, uninsured adults aged 19 to 45.
Analysis of Broader Implications
The current state of HPV vaccination in the United States is a study in contradictions. On one hand, the clinical evidence for the vaccine’s ability to prevent cancer is stronger than ever. The transition to Gardasil 9 and the inclusion of boys in the recommendation have created a framework for significant long-term reductions in cancer incidence.
On the other hand, the politicization of vaccine policy presents a new and unpredictable hurdle. The attempt to overhaul the ACIP and the subsequent legal battles have introduced a level of regulatory uncertainty that could undermine provider confidence. When clinical guidelines are perceived as being subject to political whim rather than scientific consensus, the "recommendation effect"—whereby a parent is more likely to vaccinate if their doctor strongly suggests it—is weakened.
Furthermore, the plateauing of vaccination rates suggests that the "easy wins" in public health have been achieved, and reaching the remaining 40% of the adolescent population will require targeted interventions in rural areas and communities where vaccine hesitancy is entrenched. As the legal system navigates the challenges to HHS policy, the medical community remains focused on the primary goal: ensuring that the next generation is protected from preventable, life-threatening cancers. The long-term impact of today’s policy shifts will likely not be fully understood for decades, as the latency period for HPV-related cancers means that the success or failure of current vaccination efforts will manifest in the cancer registries of the 2040s and 2050s.